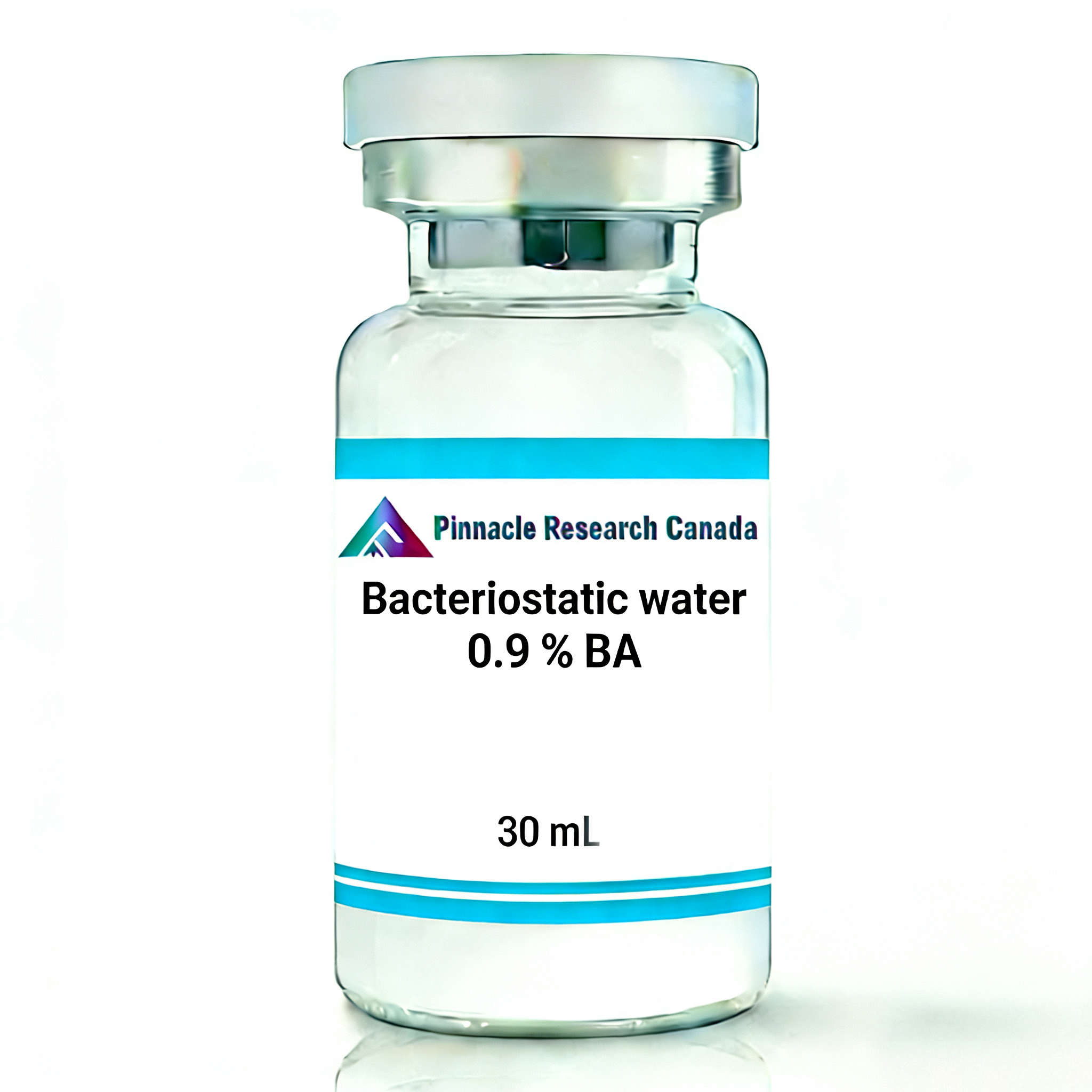
Description
Pinnacle Bacteriostatic Water for Injection – Premium Sterile Solution for Safe Peptide and Medication Reconstitution
Struggling with contaminated vials or wasted product because your sterile water expires after one use? Pinnacle Bacteriostatic Water for Injection solves both problems with a pharmaceutical-grade solution designed for the serious researcher and health-conscious individual. Unlike standard sterile water that must be discarded after a single puncture, our bacteriostatic water contains 0.9% benzyl alcohol preservative, keeping your solution sterile and safe for up to 28 days after opening. Whether you’re reconstituting peptides, HGH, HCG, or other lyophilized compounds, this medical-grade solution ensures every dose maintains perfect potency and purity—eliminating the anxiety of contamination while dramatically reducing waste and cost per use.
Key Features
- 28-Day Multi-Dose Protection: Benzyl alcohol preservative maintains sterility for nearly a month after first use, unlike single-use sterile water alternatives
- USP Pharmaceutical Grade: Meets United States Pharmacopeia standards for purity, sterility, and safety—the same quality used in medical facilities
- Perfect for Peptide Reconstitution: Optimally buffered pH and composition ideal for dissolving research peptides, growth hormone, HCG, and injectable medications
- Reduced Injection Discomfort: Properly balanced solution minimizes burning or stinging at injection sites compared to unbuffered alternatives
- Cost-Effective Multi-Use Format: One 30mL vial reconstitutes multiple products, significantly reducing your cost per injection
- Clearly Labeled & Traceable: Every vial includes batch numbers, expiration dates, and NDC codes for complete accountability
- Non-Pyrogenic Formula: Rigorously tested to be free of fever-causing endotoxins for maximum safety
- Universal Syringe Compatibility: Works seamlessly with insulin syringes, standard needles, and all reconstitution equipment
Technical Specifications
| Product Name | Pinnacle Bacteriostatic Water for Injection USP |
| Preservative Agent | 0.9% Benzyl Alcohol |
| Solution Composition | Sterile, non-pyrogenic water for injection |
| Vial Volume | 30mL per vial |
| pH Range | 5.0 – 7.0 (buffered) |
| Osmolarity | Hypotonic |
| Sterility Standard | Terminally sterilized, USP compliant |
| Unopened Shelf Life | 24 months from manufacture date |
| Post-Opening Stability | 28 days when refrigerated (2-8°C) |
| Storage Temperature (Unopened) | Room temperature (15-30°C / 59-86°F) |
| Storage After Opening | Refrigeration required (2-8°C / 36-46°F) |
| Primary Use | Reconstitution and dilution of injectable medications and research compounds |
| Packaging Type | Type I glass vial with latex-free rubber stopper |
Why Pinnacle Bacteriostatic Water is Essential for Your Research Protocol
When you invest in premium peptides, growth hormone, or other research compounds, using inferior reconstitution solutions is like buying a sports car and filling it with contaminated fuel. The quality of your Pinnacle Bacteriostatic water directly impacts the stability, sterility, and effectiveness of every compound you reconstitute.
Pinnacle Bacteriostatic water for injection Canada enthusiasts choose our pharmaceutical-grade solution because it eliminates three critical pain points:
1. Contamination Risk: Every time you puncture a vial of standard sterile water without preservatives, you introduce potential contamination vectors. Our benzyl alcohol preservative creates an antimicrobial environment that inhibits bacterial growth between uses.
2. Product Waste: Single-use sterile water forces you to discard unused portions or risk contamination. With 28-day stability after opening, our bacteriostatic water lets you use exactly what you need, when you need it.
3. Injection Comfort: Poorly buffered solutions can cause significant burning and discomfort at injection sites. Our properly balanced pH ensures smooth, pain-free administration.
For those running comprehensive research cycles, our bacteriostatic water complements perfectly with products from our peptides collection and post-cycle therapy section, ensuring every element of your protocol maintains pharmaceutical standards.
Step-by-Step Usage Instructions
Proper technique when using Pinnacle Bacteriostatic water for peptides ensures maximum safety and product longevity:
Before First Use:
- Inspect the vial carefully – solution should be crystal clear with no cloudiness or floating particles
- Check the expiration date printed on the label
- Verify the seal is intact and unbroken
Reconstitution Process:
- Sanitize: Clean the rubber stopper with a fresh alcohol swab and allow to air dry for 30 seconds
- Draw Solution: Using a sterile syringe (typically 1-3mL), draw the required amount of bacteriostatic water
- Inject Carefully: Add the water slowly to your peptide or compound vial, directing the stream down the inside wall of the glass—never spray directly onto the powder
- Mix Gently: Swirl the vial in a circular motion until powder completely dissolves (never shake vigorously as this can damage peptide bonds)
- Label Immediately: Mark both vials with the reconstitution date
Storage & Maintenance:
- After first use, store your bacteriostatic water in the refrigerator at 2-8°C
- Always use a new alcohol swab before each puncture
- Never use the same needle to both draw and inject—this introduces contamination
- Discard any remaining solution after 28 days from first use
- If solution becomes cloudy, discoloured, or contains particles at any point, discard immediately
Bacteriostatic Water vs. Other Reconstitution Options
Understanding your options helps you make informed decisions about your research protocols:
| Solution Type | Shelf Life After Opening | Best Used For | Key Limitation |
| Bacteriostatic Water | 28 days (refrigerated) | Multi-dose peptides, HGH, HCG, research compounds | Not for neonatal use |
| Sterile Water | Single use only | Large single-dose reconstitutions | Must discard after one use |
| Sodium Chloride 0.9% | 28 days (with preservative) | Medications sensitive to pure water | Not ideal for all peptides |
| Sterile Saline | Single use only | Specific medication requirements | Limited peptide compatibility |
For most researchers working with peptides and growth factors, bacteriostatic water Canada suppliers provide represents the optimal balance of safety, longevity, and versatility.
Important Safety Information
While bacteriostatic water for injection is extremely safe when used properly, please observe these critical precautions:
Do NOT Use If:
- Solution appears cloudy, discolored, or contains visible particles
- Seal or vial shows signs of damage or tampering
- Product has passed its expiration date
- Vial has been open for more than 28 days
- Product has not been stored under proper temperature conditions
Special Warnings:
- Never use for newborns or neonates – benzyl alcohol has been associated with toxicity in this population
- Always verify compatibility with your specific compound before reconstitution
- Use aseptic technique at all times to prevent contamination
- Do not inject bacteriostatic water directly – it is for dilution purposes only
- Consult manufacturer guidelines for compounds with specific reconstitution requirements
Frequently Asked Questions
How is Pinnacle Bacteriostatic water different from regular sterile water, and why does it matter?
Pinnacle Bacteriostatic water contains 0.9% benzyl alcohol as a preservative agent, which inhibits bacterial growth and allows the solution to remain sterile for up to 28 days after the vial is first punctured—as long as it’s stored properly in the refrigerator. Regular sterile water for injection contains no preservatives and is designed strictly for single-use applications. Once you puncture a sterile water vial, any unused portion must be discarded immediately to avoid contamination risks.
This difference is critical for anyone working with peptides or compounds that require multiple reconstitutions. With bacteriostatic water, you can reconstitute several vials from one source bottle over the course of a month, dramatically reducing waste and cost. For example, if you’re running a research protocol that requires weekly reconstitutions, one vial of bacteriostatic water serves your entire month, whereas sterile water would require four separate vials. Beyond economics, bacteriostatic water provides peace of mind—you know each draw is protected against bacterial contamination by pharmaceutical-grade preservation.
How long can I safely use bacteriostatic water after opening the vial?
Once you’ve made the first puncture, Pinnacle Bacteriostatic water for injection remains safe and effective for up to 28 days when stored in the refrigerator between 2-8°C (36-46°F). This 28-day window is based on extensive pharmaceutical stability studies showing that the 0.9% benzyl alcohol concentration maintains antimicrobial effectiveness throughout this period.
To track this properly, always write the date of first use directly on the vial label with a permanent marker immediately after your initial puncture. Before each subsequent use, perform a visual inspection—the solution should remain perfectly clear with no cloudiness, color change, or floating particles. If you notice any changes in appearance, discard the vial immediately regardless of how much time has passed.
It’s equally important to maintain proper storage discipline. Always return the vial to refrigeration immediately after each use, never leave it at room temperature for extended periods, and always sanitize the rubber stopper with a fresh alcohol swab before each needle insertion. Following these protocols ensures you get the full 28-day benefit while maintaining pharmaceutical-grade sterility. After 28 days, even if the solution looks perfect, the preservative effectiveness diminishes and the vial should be replaced.
Can bacteriostatic water be used with all types of peptides and injectable research compounds?
Pinnacle Bacteriostatic water is compatible with the vast majority of peptides, including popular research compounds like BPC-157, TB-500, Ipamorelin, CJC-1295, and many others, as well as HCG, HGH, and various lyophilized medications. Its neutral pH balance and simple composition make it the universal reconstitution standard in research and clinical settings.
However, there are important exceptions to be aware of. Some specialized compounds come with specific manufacturer reconstitution instructions that may require sterile water without preservatives, sodium chloride solution, or other specific diluents. This is particularly true for certain sensitive peptides or medications where benzyl alcohol could potentially interact with the active compound or reduce stability. Always consult your product’s technical documentation or manufacturer guidelines before reconstitution.
Additionally, Pinnacle Bacteriostatic water should never be used for neonatal applications (newborns under 28 days old) as benzyl alcohol has been associated with serious adverse reactions in this population. For general research purposes with standard peptides and growth factors, bacteriostatic water from Canadian Anabolic Source provides the ideal reconstitution solution—just verify compatibility with your specific compound first. When in doubt, reach out to our knowledgeable support team or consult with a qualified healthcare professional familiar with your research protocol.
Maximize Your Research Results with Pinnacle Bacteriostatic Water
Your research compounds represent a significant investment in your goals—whether that’s cutting-edge peptide research, optimized recovery protocols, or advanced health optimization. Don’t undermine that investment with substandard reconstitution solutions that introduce contamination risks, waste product, or compromise peptide stability.
Our pharmaceutical-grade Pinnacle Bacteriostatic Water provides the professional-level quality you need for consistent, reliable results. With 28-day multi-dose capability, USP-compliant manufacturing, and rigorous quality testing, you get the same sterile solution trusted by medical facilities and research laboratories across North America.
Canadian Anabolic Source is committed to providing not just products, but complete research solutions with:
- Discreet, secure shipping across Canada
- Pharmaceutical-grade quality assurance on every product
- Knowledgeable customer support for technical questions
- Complete ancillary product selection for comprehensive protocols
- Competitive pricing without compromising on quality
Stock your research facility with the reconstitution standard trusted by serious researchers. Order your Pinnacle Bacteriostatic water today and experience the difference that pharmaceutical-grade quality makes in every injection.
Disclaimer: This product is intended for research purposes only. Bacteriostatic Water for Injection should be used by qualified individuals following proper aseptic technique and safety protocols. Always consult with a healthcare professional regarding appropriate usage, storage, and compatibility with specific compounds. This product is not intended to diagnose, treat, cure, or prevent any disease.





Reviews
There are no reviews yet.